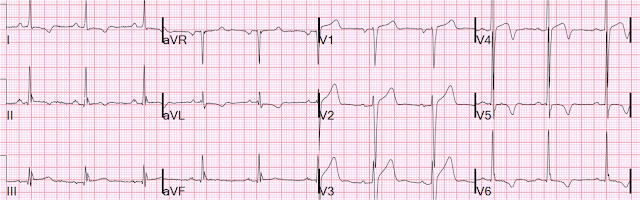
A 30 year old African American Male presented to the ED with chest pain that occurred the day before. It had been 6-7/10 in intensity and lasted for about 10 minutes, and was associated with strenuous activity. It radiated to the left arm and was associated with SOB. There was no pain on the day of presentation.
Here is the ED ECG:
Since this is a young (30 years old!) patient, so it can't be acute MI, right? And the patient is African American, so it must be one of those normal variants in blacks, right?
Wrong!!See our article: Walsh, B., Macfarlane, P. W., Prutkin, J. M. and Smith, S. W. (2019) Distinctive ECG patterns in healthy black adults. Journal of Electrocardiology, 56, pp. 15-23. (doi:10.1016/j.jelectrocard.2019.06.007)
(Full text here: https://eprints.gla.ac.uk/189824/7/189824.pdf)
The ECG above is diagnostic of Wellens' syndrome (full reference below):
1) Episode of anginal chest pain that is resolved (GONE!)
2) preserved R-waves
3a) Terminal T-wave inversion (biphasic T-waves). This is Pattern A.
3b) Deep symmetric T-waves (Pattern B)
What does it mean? ----It is a reperfusion pattern!!
It means that the patient had full occlusion at the time of the chest pain, but that there was spontaneous reperfusion ("recanalization") resulting in resolution of pain and what I call "reperfusion T-waves"
That these T-waves are due to reperfusion was published by Wellens' himself (as senior author) a decade after he described Wellens' waves.
Wehrens XH, Doevendans PA, Ophuis TJ, Wellens HJ. A comparison of electrocardiographic changes during reperfusion of acute myocardial infarction by thrombolysis or percutaneous transluminal coronary angioplasty. Am Heart J 2000;139(3):430–6.
Doevendans PA, Gorgels AP, van der Zee R, Partouns J, Bar FW, Wellens HJJ. Electrocardiographic diagnosis of reperfusion during thrombolytic therapy in acute myocardial infarction. Am J Cardiol 1995;75(17):1206–10.
Yet very few know about this.
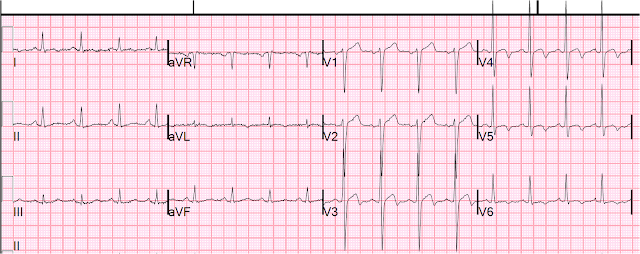
But the PMCardio Queen of Hearts AI Model knows that this is a reperfused OMI:
Here is a demonstration (Case 2):
First ED ECG is Wellens' (pain free). What do you think the prehospital ECG showed (with pain)?
When the patient had chest pain, prior to nitroglycerine, what do you think the ECG showed?
Here is the prehospital ECG, recorded in the presence of pain:
 |
| Hyperacute anterolateral STEMI |
The medics had activated the cath lab and the patient went for angiogram and had a 95% stenotic LAD with TIMI-3 flow. A stent was placed. The peak troponin was only 0.364 ng/mL (equivalent to 364 ng/L). So even a massive STEMI, if it reperfused quickly, can result in a relatvely low troponin (in contrast to the next case!)
Here is a case that shows how Wellens' pattern evolves from a subtle OMI (Case 3):
Paramedics make a great call
A middle-aged male called 911 for chest pain.
Here was the first prehospital ECG with pain at 5/10:
 |
| Computerized QTc is 418 ms. There is nondiagnostic ST elevation in V1-V4. If you use the 3 variable formula, you get 25.15, (> 23.4, which is all but diagnostic of LAD occlusion). The medics did not use the formula, as far as I know. Now we use the 4 variable formula |
Or, better yet, the PMCardio Queen of Hearts AI app:
Medics were worried, and gave nitroglycerine, then repeated the ECG at 5 minutes with pain at 2/10:
 |
| Less ST elevation |
And they repeated again with pain at 1/10 at 9 minutes:
 |
| Near Normal |
Medics asked for physician interpretation on arrival. Physicians were worried and activated the cath lab. The first (and only) ED ECG is here:
 |
| QTc 386. Most ST elevation is resolved. Formula value is now down to a very low value of 19.352 |
A 90% thrombotic LAD lesion was found and stented. There was pre-procedure TIMI-3 flow (perfect flow)
Door to balloon time was 25 minutes.
Peak troponin I was 17 ng/mL (this is quite a large infarct).
IMPORTANT: notice that an LAD Occlusion can have TIM-3 flow at angiogram because it opens spontaneously, AND that the troponin is often very high and indicative that the artery was indeed occluded at the time of the first ECG.
Subsequent Echo showed EF of 56% and distal septal, anterior, apical, and
 |
| You can see the beginning of terminal T-wave inversion in V2 and V3. Had there been no prior ECGs, this patient who is now pain free would be suspected of Wellens' syndrome |
 |
| Evolving T-wave inversion, classic Wellens pattern B morphology |
 |
| Full blown Wellens' Pattern B terminal T-wave inversion. |
6. Finally, Transient STEMI should be taken emergently to the cath lab. Failure to do so can result in Disaster: Spontaneous Reperfusion and Re-occlusion - My Bad Thinking Contributes to a Death
PseudoWellens Patterns: all of the below were recorded in patients with active pain (which is reassuring!)
What is this strange looking ECG in a young woman?
Is it important to recognize LVH Pseudo-infarction patterns?
LVH
Pseudo-Wellens' Syndrome due to Left Ventricular Hypertrophy (LVH)
LVH
Finally, Wellens Pattern A evolves over time to Wellens Pattern B. See this amazing series of ECGs over time:
Classic Evolution of Wellens' T-waves over 26 hours
MY Comment, by KEN GRAUER, MD (4/2/2025):
- As per Dr. Smith — the fact that the patient whose ECG is shown in Figure-1 is a young adult African American male who presented with short-lived chest pain (lasting only ~10 minutes) the day before — is potentially as misleading as can be, given that the overwhelming majority of times that this type of history will be associated with some form of repolarization variant that is so commonly seen with this demographic. But not this time!
- The diagnosis of Wellens' Syndrome is clearly made more difficult by the presence of LVH. This is not to say that Wellens' Syndrome cannot occur when LVH is present — but rather that assessment of ST-T wave abnormalities may be complicated if there are ST-T wave abnormalities as a result of LV "strain". That said — although the S wave in lead V2 measures 21 mm — R wave amplitudes in the lateral chest leads of ECG #1 are modest, such that voltage criteria for LVH are not met (For quick review of ECG criteria for LVH — Click on the Tab in the lower row of the Menu at the top of each page in this ECG Blog). Given the young age of today's patient ( = 30 years old) — even greater amplitudes are required to satisfy LVH voltage criteria than those cited in the above Menu bar.
- A similar pattern of T wave inversion is seen in no less than 5/6 of the chest leads. ST-T wave changes of Wellens' Syndrome are most characteristically seen in leads V2,V3,V4 — but less commonly in leads V1 and V5 (as seen in today's case).
- The very straight and steep T wave descent (highlighted by the slanted RED lines that are best seen in leads V2 and V3). This generally is not seen with LV "strain" or repolarization variants.
- Straightening of the ST segment takeoff in leads V3 and V4 (double RED arrows in these leads) — with an unusually wide base to the T waves in these leads.
- Lack of many criteria cited by Drs. Smith and Meyers as characteristic of BTWI (Benign T Wave Inversion) — including: i) Lack of prominent J-point notching that is characteristic of repolarization variants; ii) Greatest T wave inversion in V2,V3,V4 instead of V5,V6 as is seen with BTWI; iii) Lack of ST elevation in most of the leads with T wave inversion (as is typically seen with BTWI); iv) Lack of tall R waves in the lateral chest leads; — and, v) Lack of T wave inversion being also seen in the inferior leads (See My Comment at the bottom of the page in the June 30, 2023 post for review of these characteristic findings with BTWI).






-USE.png)





.png)

-USE.png)



-USE.png)